11. System Specifications and Symbol
Specifications
| Item | Value |
| Dimensions of TrakStation (L x W x H) | Varies by model of tablet computer, typical dimensions based on 10” screen size.
25.0 cm x 17.5 cm x 0.9 cm (9.8 in x 6.9 in x 0.3 in) |
| Weight of TrakStation | Varies by model of tablet computer, typical dimensions based on 10” screen size. |
| Minimum External connections |
DC-in jack |
| Touchscreen | 10” to 12” touch screen (1280 x 800 or better) |
| Power requirements | |
| TrakStation AC power adapter | UL marked power adapter Input to power adapter: 100 – 240 V50-60 Hz Output (varies by model of tablet computer): 5 to 19.5 V DC, 2 to 4 A |
| USB Hub AC power adapter | UL marked power adapter Input to power adapter: 100 – 240 V AC, 50 – 60 Hz 1.2 A Output: 12 V DC, 4 A |
| Power requirements | |
| TrakPod power | Input: DC power over USB connection 5 V DC, 0.5 A (standard USB 2.0) |
| Operating, Transport and Storage temperature | 5 – 40°C (41 – 104°F) |
| Operating, Transport and Storage humidity | 5-95% relative humidity, non-condensing (For the components outside of an incubator) |
| Operating environment | Intended for indoor use only Altitude not specified, Pollution Degree 2 |
| Operating, Transport and Storage humidity | 5-95% relative humidity, non-condensing |
TrakStation Performance Specifications
| Item | Value |
| pH measurement accuracy and range | 0.05 pH units within pH 7.00 to 7.60 |
| pH resolution | 0.01 pH units |
| pH Sensor lifetime | 7 days of use with 30 minute reading frequency. 3 days of use with 1 minute reading frequency. Shelf life is labeled on the packaging of sensor. (Typically one year from pHID assignment) |
| Optical control | QC Alignment Tool (minimum 12 month lifetime) |
Certification
| Item | Value |
| Safety class | Class A |
| FCC | Part 15 Subpart B Compliant |
| 2014/30/EU | Electromagnetic compliant |
| 2014/35/EU | Low voltage compliant |
| EN 61010 revision 3 | Conforms |
| RoHS | Compliant |
Specifications are subject to change without notice.
Symbol Glossary
Symbols used in all labeling are summarized below in the table.
| Symbol | Symbol Title | Symbol Description |
| Caution | Indicates a potentially hazardous situation which, if not avoided, may result in minor or moderate injury. It may also be used to alert against unsafe practices. | |
 |
Manufacturer | Indicates the device manufacturer. |
 |
Authorized Representative in European Community | Indicates the Authorized representative in the European Community. |
 |
Date of Manufacture | Indicates the date when the device was manufactured. |
 |
Use-by Date | Indicates the date after which the device is not to be use. |
 |
Batch Code | Indicates the manufacturer’s batch code so that the batch or lot can be identified. |
 |
Catalogue Number | Indicates the manufacturer’s catalogue number so that the device can be identified. |
 |
Serial Number | Indicates the manufacturer’s serial number so that a specific device can be identified. |
 |
Keep Away From Light | Indicates a device that needs protection from light sources. |
 |
Sterilized using ethylene oxide | Indicates a device that has been sterilized using ethylene oxide. |
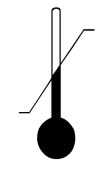 |
Temperature Limit | Indicates the temperature limits to which the device can be safely exposed. |
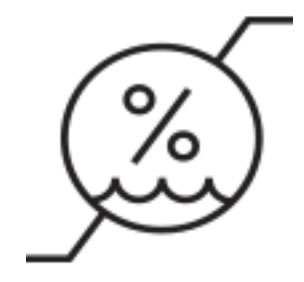 |
Humidity Limit | Indicates the range of humidity to which the device can be safely exposed. |
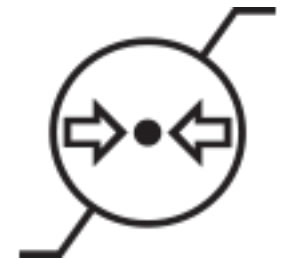 |
Atmospheric Pressure Limitation | Indicates the range of atmospheric pressure to which the device can be safely exposed. |
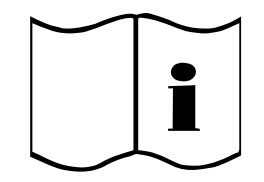 |
Consult Instructions For Use | Indicates the need for the user to consult the instructions for use. |
 |
Control | Indicates a control material that is intended to verify the performance characteristics of another medical device. |
 |
CE Marking | Signifies European technical conformity. |
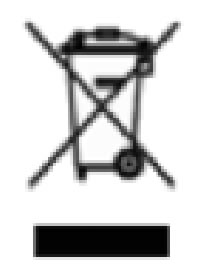 |
Recycle Electronic Equipment | Do Not Dispose of this equipment in normal trash. |
 |
FCC Declaration of Conformity | Certification mark employed on electronic device which indicates that EMC is under limits approved by FCC. |
Intellectual Property
“SAFE Sens”® , “TrakStation”® , “TrakPod”® , and “Innovations that make a difference”® are registered US, Canadian and/or European trademarks owned by Blood Cell Storage, Inc. (BCSI).
The SAFE technology is covered by multiple issued and pending US and International equivalent Patents including but not limited to: US Pat. No. 7,608,460, 7,968,346, 8,148,167, 8,183,052, 8,497,134, 9,040,307, 9,062,205, 9,217,170; AU Pat. No. 2005277258; JP 特許番号 5017113; CN 专利号 ZL200580033961.1; IN Pat. No. 257667.
